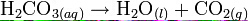a. Analyze and interpret data on the properties of substances before and after the substances interact to determine if a chemical reaction has occurred.
Physical properties and physical changes of substance
|
Physical properties are qualities that is can be observed without changing the sample's composition. Characteristics such as color, texture, smell, taste, appearance,... are physical properties.
Example - Glass has the property of being clear and hard in solid form. Physical changes are changes in the substance in which it only alters the appearance but does not change its composition. Changes in shape, color, temperature, forming precipitation gas are physical changes. Example - When putting ice into a glass of water, the ice is melted but the substance still has the same composition. |
Chemical properties and chemical changes of substance
|
Chemical properties are characteristics that give the substance ability/inability to undergo a change that alters its composition. Some examples are ability to burn, ability to react with a certain substance, inability to react with a certain substance,...
Example - Paper's ability to burn is a chemical property because with burning, the paper's overall composition is changed and turn into ash. Chemical changes are changes in which the composition of the substance is altered by a reaction. Changes such as rusting, burning (combustion), changing into a new substance,... Example - Rusted iron undergoes a change in which the iron is oxidized (chemically interact with oxygen), eventually become iron dioxide. |
b. Develop and use a model to describe how the total number of atoms does not change in a chemical reaction and thus mass is conserved.
Chemical reaction
|
A chemical reaction happens when the composition of 2 or more substance is rearranged at molecular level. The overall amount of mass and all the atoms present before the reaction remain after the reaction has occurred.
Example - In the process of cellular respiration, glucose reacted with oxygen, resulting in different substances with altered composition (with extra energy released) but all matter presented before the reaction is still present after the reaction. |
The Law of Conservation of Mass
|
The Law of Conservation of Mass states that in a chemical reactions, atoms that involved are neither created nor destroyed. All atoms present in the reactants are also present in the products. The chemical equation are always balanced.
Example - In a soda can, if being left open too long, a chemical process will happen in which the carbonic acid (H2CO3) are decomposed into water (H2O) and carbon dioxide (CO2). In this chemical equation, it is balanced, since all atoms of the reactant are only rearranged, and no atoms are being created nor destroyed. |
c. Undertake a design project to construct, test, and modify a device that either releases or absorbs thermal energy by chemical processes.
Exothermic reactions
|
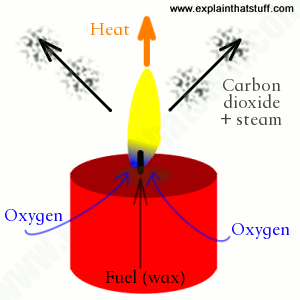
In a chemical reaction, if more energy is released when the products formed than the energy that needed to break the bonds between the reactants, that is an exothermic reaction. An exothermic reaction is a reaction in which energy is released in the form of heat or thermal energy. Combustion is a typical form of exothermic reaction, in which the reactants are usually Oxygen and Carbon-Hydrogen based substance.
Example - In candles, the wax, which is a carbon-based material, reacted violently with the Oxygen in the air, and released so much energy that it created light, or fire and other byproducts such as steam and carbon dioxide. By measuring its surrounding (the air), it can be clearly seen that the reaction has released energy into its environment. |
Endothermic reactions
|
In a chemical reaction, if more energy is required to break the bonds between the reactants than is released, that is an endothermic reaction. That is, the reaction absorbs energy. An endothermic reaction is a reaction in which energy is absorbed.
Example - An ice cube that melts absorb the thermal energy of its surrounding in order to turn from a solid to a liquid. The energy is absorbed in order for the molecules to move past each other, forming a liquid. With this reaction, by measuring its surrounding, we can easily see that it has absorbed thermal energy from its environment in order to move from a solid to a liquid. |