a. Develop models to describe atomic composition of simple molecules and extended structure.
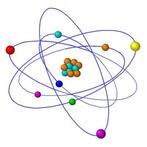
AtomsAn atom is the smallest unit of matter. All atoms are created from the 3 main particles: proton, neutron and electron. Protons and neutrons together formed the nucleus, while the electrons formed the outer layers. Atoms consist mostly of empty space, since the distance between the nucleus and the electrons are huge, ratio wise.
Example - Atoms make up every form of ordinary matter. |
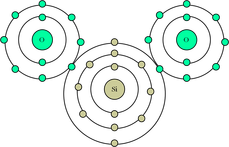
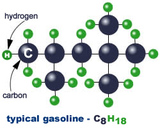
Simple Molecules
Polymers/Extended Structrue
b. Develop a model that predicts and describes changes in particle motion, temperature, and state of pure substance when thermal energy is added/removed.
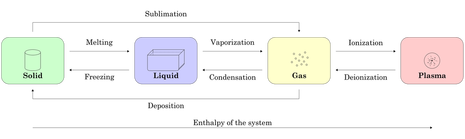
States of matter - Solid, Liquid, Gas and Plasma
Solid
|
In solid form, with little energy, atoms are fixed in a crystal structure depend on the substance. Molecules vibrate in place. This state can be achieve by removing thermal energy from the state of liquid or gas.
Example - Metals like iron are in solid form. The molecules are arranged in a fixed position while vibrating in place. |
Liquid
|
In liquid form, with more energy, molecules slide past each other. The substance takes the shape of its container and have a fixed volume. This state can be achieve by adding energy into the solid state or removing energy from gas state.
Example - The water that people drink everyday are in liquid form. Molecules in water slide past each other. |
Gas
|
In gas form, with a higher level of energy than liquid, molecules are spread in every direction. Substances do not have a fixed volume or shape, therefore take the shape and volume of its container. This state can be achieve by adding thermal energy into solid or liquid state.
Example - The oxygen that humans breath everyday are in gas state. Oxygen molecules are spread in every direction. |
Plasma
|
In plasma form, atoms break apart to form individual nucleus and electrons. With so much energy, the molecules collide strong enough to break apart. This state can be create by adding significant amount of thermal energy into the gas form of the substance. Plasma is also known as the hottest state a substance can achieve.
Example - The neon lights are the plasma state of the element neon. |